News/Photo
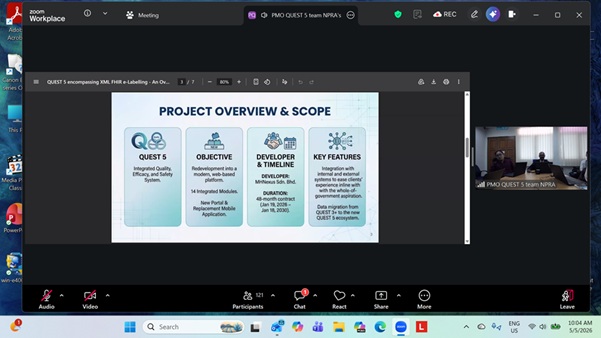
E-Labelling sharing session: From PDF to FHIR ready XML
On 5 May 2026, MAPS attended webinar on From PDF to FHIR ready XML for E-labelling. Topic discussed includes:
- FHIR & PI fundamental
- Proposed implementation process flow
- Country specific (Jorddn) experience: Challenges, Mitigation & Readiness.
- Malaysia E-labelling implementations & NPRA directions.
MHNesus Sdn bhd has been awarded to develop the Quest 5 system which is XML PI compatible.
Technical Committee Meeting on Medicines and Medical Devices security Number 1/2026
The meeting was chaired by Dr. Azuana Binti Ramli, Deputy Director General of Health (Pharmaceutical Services) at Delima meeting room, Pharmacy Services Program, Jalan Universiti. It was held on 30 April 2026 from 2.30 to 5 pm.
Feedback was provided from all the industry representatives of pharmaceutical (MAPS, MOPI and Phama) and medical devices industries. In the meeting it was revealed that pharmaceutical supplies were still adequate, although prices are facing pricing challenges. Only about a dozen pharmaceutical products may face some supply challenges.
Global Supply Crisis: Can Healthcare Sytems Cope
Astro Awani engaged with the industry on the subject in the program "Consider This". The broadcast was on 27th April 10 pm. Click on the picture below to watch the broadcast.

Dialogue with Pharmaceutical Service Program on impact of Gulf Conflict
This meeting on 27th April, chaired by Pn Nur Ain Suhaila Binti Shohaimi as Director (Pharmaceutical Policy & Strategic Planning Division) focused on discussing supply chain challenges and price increases in the pharmaceutical industry due to ongoing conflicts and logistics issues. Company representatives from various pharmaceutical firms, including Medispec, Pharmaforte, Apex Pharmacy, and others, shared concerns about API shortages, increased freight costs (20-30%), and delayed deliveries affecting both tender items , Lampiran Q and local purchase orders. Key issues discussed included suppliers from Bangladesh and Thailand requesting 40% and 15% price increases respectively, suppliers from India facing delays due to packaging material shortages, and the need for forward-dated purchase orders to better manage inventory planning. The meeting also highlighted transportation challenges within Malaysia, including fuel shortages affecting outbound deliveries from warehouses, and the potential impact on pricing due to rising logistics costs
Next steps
PSP Pn Nur Ain to
- Discuss with Amalan team about the possibility and process for issuing forward/advance POs (for up to 6 months) for tender and Lampiran Q products, and provide feedback
- Engage with NPRA to facilitate/prioritize approval for alternative suppliers (as per Apex Pharmacy's request and potentially others)
- Review and consider requests for exemption from penalties on a product basis if companies submit such requests
- Facilitate discussions with relevant authorities to expedite awarding of KKM contracts where possible
- Share contact/email details (ain.shuhaila@moh.gov.my) with participants for submission of product lists and concerns
- Raise the issue of fuel supply prioritization for essential industries (including pharmaceutical logistics) at higher government levels
Collaboration
- All companies can email Pn Nur Ain (ain.shuhaila@moh.gov.my) team with details of specific products (including tender/LPO/Lampiran Q items) that may face supply or price issues, so MOH can facilitate and discuss with relevant teams
MOPI Webinar 3-2026: Patent Linkage Implementation under CPTPP
On 16 April 2026, Third World network (TWN) presented an overview of the relevant CPTPP provisions and the proposed Patent Linkage obligations in Malaysia as well as a discussion on the potential implications of the proposed framework on access to generic medicines and broader public health considerations. Key issue is:
- Eligible patent : too broad coverage for all patent.
- Not clear in the guideline if registration process will be halted in the 12 months suspension period or registration process ongoing as per verbally informed to industry on 11 Mar 2026
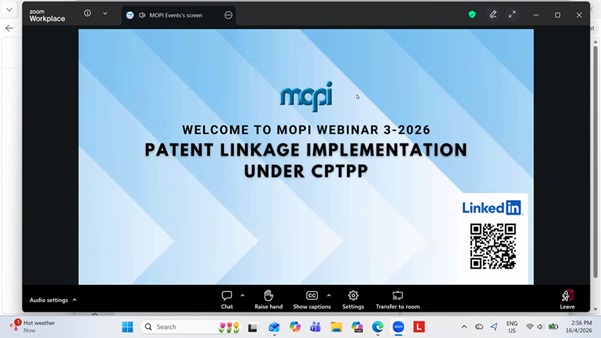
Preparing for Pharmaceutical Track & Trace in Malaysia
On 8 April 2026 9am, webinar on preparing for pharmaceutical tract and trace was held with presentation from GS1, MOH and various industry players. MOH tentatively aiming to launch mandatory for all poison items wef Q3 2028. Slides have been sent out to all MAPS RAs via emails.
Annual General Meeting 2026
On 2 April 2026, MAPS held its 15th AGM with 58% quorum. 22 out of 38 members turned up for the AGM. Various issues were discussed mainly on regulatory matters. The BOD informs the coming 5th MAPS dinner will be held on 20 Nov 2026 (Friday). The next AGM will be held at Medispec new building. Address to be distributed later.
Dunas Meeting Component 1
On 30 Mar 2026, JKT Dunas Componant 1 on Governance in medicine was held in hybrid, chaired by Pn Nur Ain Shuhaila binti Shohaimi at 9am.
- 4 strategies, 6 initiatives and 12 indicators were presented with 92.3% achievements reached.
- Garispanduan on credentialing for registered pharmacist uploaded to portal pharmacy. gov.my.
- Module GGM for private sector ongoing with dialogue held on 24 Feb 2025.
- No payment or charges required for renewal of credentialing.
- 99.32% adoption by MOH Hosp, PKD, & KK adopted the guidelines.
- Strethening of SDO 1952, discussion of the draft with AGC through 3 discussion sessions. Tabled in June 2026 in parliament.
- ROPA 1951 delay. Focus on SODA 1952.
- MASA 1956, finalizing & drafting the proposal paper for MOH top management approval by Q2 2026.
- Altogether 9 credentialing modules for pharmacists approved.
- a total of 55 MOH RX credentiated, 19 Pharmacy assistant being credentiated.
Discussion on Medicine status caused by Middle East Conflict
The Pharmacy services program called an emergency meeting on 26th March to discuss the status of medicine security and to pave the way forward in preventing shortages and to handle the potential spike in prices.
The Deputy Director-General (Pharmacy Services) YBrs. Dr. Azuana binti Ramli of the MOH chaired the meeting which was attended both on-site and on-line by many representatives of the industry and affected downstream stakeholders (Military, teaching hospitals).
We thank all members for the feedback rendered to us very quickly, and Dharshen presented our findings to the attendees.
The priority would be to ensure supplies of essential and single source medicines. Concerns over escalation of costs in direct and indirect consequences of fuel prices were also discussed. A multi-party task force of stakeholders for purpose of monitoring this situation will be set up, and the draft terms of reference has been prepared.
We urge all members to co-operate with the Pharmacy Services Progam in their information gathering when the mechanism is set up, and assurances of confidentiality has been given. Let us work together to ensure that our nation makes it through this difficult time.
E-labelling committee meeting Bil 1 2026
On 12 Mar 2026, JK E-labelling Bil 1 2026 was held hybrid chaired by Pn Wan Noraimi Binti Wan Ibrahim
- E-labelling surveys on the effectiveness and perception of Public, HCP and Industry/NPRA was presented by Dr June Choon (Sunway University). Age is the limiting factor to using E-labelling. 82% of age >60 are less likely to use E-labelling.
- The use of XML to replace PDF format for PI was presented by Mr Kobu, to start with Quest 5 live 1 implementation w.e.f Sept 2027.
- DHCP letter to be omitted after agreement obtained from PBKD in April 2026
2nd Engagement Session on Implementation of Patent Linkage
On 11 Mar 2026 9am to 11.30am, a second dialogue on patent linkage was held and chaired by YBrs Pn Wan Noraimi binti Wan Ibrahim.
- Purpose is to inform all stakeholders on the latest updates of the patent linkage guidelines, explained operational procedures of Quest 3+.
- Patent linkage applies to all pharmaceutical products but excludes biologics.
- Only applies to product patents and indication patents. It excludes manufacturing process patent, formulation patents, polymorphs, salts/esters, dosage form and packaging patents.
- Category 1,2, 3 products need not go through patent linkage.
- Notification Only applies to category 4 products, whereby it may infringe patents, notifications 45 calendar days, Acknowledgements 20 days, Suspension up to 12 months if there's litigation.
- UPC will be held from 1 to 30 April for industry to provide feedback
Dunas Meeting Component 2
On 31 Jan 2026, DUNAS (Quality, Safety & Efficacy of medicines) was held at 9am, chaired by Pn Rosliza Binti Lajis, Deputy director of NPRA.
- Sales of drugs (medicinal product for clinical trial) SODIP approved on 28 Mar 2025. UPC concluded from 17 Oct 25 to 17 Nov 25. Subject to SODA amendment approval by parliament, schedule to be tabled in June 2026. If things proceed smoothly Act to be gazetted in Oct 2026.
- WHO maturity level 3, non-legal IDP 65, Legal IDP 19. To achieve benchmarking by WHO ML3 by 2026.
- Tabung Amanah Latihan (L720) set up for Donation to achieve ICH membership. However industry donation need to subject to Jawatankuasa Akaun Amanah for approval since there maybe conflict of interest.
- Cell & Gene Therapy Registration. Survey conducted on registration barriers to the approval of CGT in Malaysia. Research ended Mar 26. The applications for GMP (8 applications), CTIL/CTX (2), Reg application (1), Reg approval (1) (Zolgensma)
- NPRA as statutory body, MOF disagreed, proposed to change to Autonomous Body instead. Task force formed on 11/9/2025 with Zaid Ibrahim & co invited to assist.
- Track & Trace. Tender contract awarded to Netsmart Sdn Bhd on 1 Oct 25 for a duration of 6 years, to set up repository. Q2 26 to Q2 27 to set up PTTS, Q2 27 to Q2 28 will include industry. There are complaints on the quality of the new hologram and on difficulty to scan using MyUbat. Duopharma shared 4 to 5 counterfeit products detected last year. Important to set up track & trace to ward off counterfeit medicines.
- Clinical trial on First in Human Study. 2 applications done locally. Current site: SUS & Ampang. Site submitted for approval: UMMC, Sunway, UITM.
- E-labelling. 1011 Products approved. [biologics 52, NDP 290, Generics 625, OTC 44]
- Combating online sales of illegal & falsified medicines 98.54% reduction. Removed 14641, caught seller 522, enforcement achieved 99.57%.
- PV inspection 9 PRHs inspected. Draft guideline to be fine-tuned.
- FRP. NCE 27, Biologics 17 Generics 12. Singapore as reference country maybe only for NCE because generics do not have evaluation report.
Dunas Meeting Component 3
On 29 Jan 2026, JKT DUNAS component 3 on Access of Medicine was held in hybrid and chaired by Pn Zuhaini binti Mukrim, Director of Bahagian Amalan & Perkembangan.
- MOPI presented predatory pricing by MNC affecting local generics launching. Duopharma spent RM30M + RM2M in CAPEX and R&D on Trevive, but Norvartis drop prices of Glivec fr RM6000 to RM 1000. Similarly, Roche drop prices of Herceptin from RM6000 to RM3000. PhAMA mentioned the short timeframe of 5 to 6 years only to market in Malaysia before losing patent. Solution from A&P: to award tender to 2 products to get maximum benefits. MAPS mentioned one of the solutions is to shorten registration timeframe. NPRA responded Quest 5 and future AI evaluation will shorten the time of registration
- Draft formulary TCM has been approved in Oct 2025. Directive will be sent out soon
- Medicine shortage Google form implemented. It will be mandatory by 1 July 2026. Guideline on reporting of medicine shortage and discontinuation already uploaded to NPRA website
- A&P noticed many medicines increase their prices comparing from KPK approval to registered product. APHM cited no competition leads to that. MAPS mentioned the need to meet registration requirements like Zone 4B and BE pushed said drug prices higher. Consensus: quality and safety still the priority and registered drugs ensure regular availability of medicines
- No initiative yet to do Drug pricing control. Even setting ceiling price is deemed not feasible, as it will affect accessibility of Medicines
- Pharmacoeconomic framework is mandatory for listing into Drug formulary
- Guideline for submission of dossier for listing into the MOH Formulary to be implemented before Dec 2026
- To shorten timeline of accessibility of new medicine, PhAMA suggest doing concurrent registration and blue book listing process
- 8th NEML will be revised and published in 2026
- 15 generics granted priority review approval
- MH Nexus awarded with Quest 5 tender and SST signed on 2 Jan 2026
- Pool Procurement of Public and Private framework developed but deem this initiative not able to be carried out due to inability to guarantee benefits to the public
- Joint API stockpiles proposed instead by Malaysia Productivity Corporation
- Price transparenvy mechanism to display prices at private sector faced objection from Drs and Dental Associations. Court grants interim suspension to this initiative
- National Generics Medicines and Biosimilars framework developed in June 2025
- Objective is Affordability and accessibility of medicine with enhanced competition to manage price escalation of medicines
- 5 strategies, 15 Initiatives and 17 Indicators with 70.58% achieved.
Dunas Meeting Component 5
On 27 Jan 2026 2.30pm, Dunas Component 5 on Partnership & Collaboration for the healthcare Industry meeting was held and chaired by Ybrs Pn Wan Noraimi binti Wan Ibrahim, Director of NPRA.
- Objective is to discuss the progress of 8 Strategies, 9 initiatives and 12 indicators, whereby % of achievement is 83%.
- Pilot project with Alpro Pharmacy on safe medicines disposal has been completed. Awaiting data publication.
- Outsourcing of patient follow up Med supply from MOH facilities to community pharmacies pilot project in Penang ended. The fees are borne currently by MOH. For phase 2, fees (management, reimbursement and possible pool procurement) yet to be finalized. Perhaps can work with state governments. This pilot project involved 2000 patients. Duration of pilot is one year until 24 Dec 2026.
- ADSSR plan of action presented during ASEAN meeting in 22 to 25 Sept 2025. 9 activities agreed upon. ADSSR framework to be drafted in 2026. My MedSecurity is one of the initiatives.
- Established My MediSafe blueprint. Launched on 25 Sept 2024.
- 3 private laboratories recognised to conduct assay and tests on traditional active ingredients.
- Feasibility study conducted on sending e-prescription from MOH facilities to the private sector. Currently using Mysejahtera and MyUbat Apps. However, only facilities using the same Pharmacy Information System (PHiS) can access.
- Track and trace tender has been awarded.
- June 2026 Bengkel DUNAS will be held.
Dunas Meeting Component 4
On 21 Jan 2026 9am to 10:30am, Ybrs En Mohd Zawawi bin Abdullah, Director of Enforcement division chaired the meeting on DUNAS (Dasar Ubat Negara) Component 4 on Quality use of medicines. Overall achievement is 87.76%
- Moving forward will invite Shopee, Lazada and Tik Tok to handover declaration on ToBAT (Tolak Ubat Tidak Sah) in conjunction with National Regulatory Conference in July 2026
-
Braille products currently not in tender specification. However, PRH can put braille as a value-added benefit during tender submission
-
Draft on Good Dispensing Practice on the way
-
Amendment on sales of drug act to be tabled to parliament in July 2026.
MyCEB and RSE Meeting with MAPS
On 12 Jan 2026 at 2pm, MAPS met up with personnel from MyCEB (Malaysia Convention & exhibition Bureau) and RSE (Reed Sinopharm Exhibitions) on possible collaboration, especially during the below events:
- tHIS (The Health Industry Series) for Asean participants dated 28-30 July 2026 at KLCC Convention Center. Organizer: RSE + APHM.
-
PharmChina dated 13-15 May 2026 Shanghai with 4500 distributors and 2000 exhibitors. The biggest exhibitions for pharmaceuticals in China
- China International Medical Equipment Fair dated 9-12 April 2026 at Shanghai with 5000 exhibitors.
We discussed on the challenges met with China manufacturers mainly in:
- Regulatory hurdles. (GMP inspections , Zone 4B, BE etc)
- Volume hurdles. (Expectation and batch size too big for Malaysia market)
Click for more info on tHIS 2026 and Reed Sinopharm.

View Archived News
Oct 2024 - Dec 2025
Nov 2023 - Sep 2024
Jan 2023 - Oct 2023
Jan 2021 - Dec 2022
Jan 2018 - Dec 2020
Aug 2016 - Dec 2017
Dec 2014 - Jul 2016
Jul 2014 - Nov 2014
Jul 2013 - June 2014
Jan 2013 - June 2013
July 2012 - Dec 2012
June 2012 and earlier
|
